
|
| Figure 1: Apple and seeds. Seeds of the common apple contain the ingredients for producing hydrogen cyanide as a mechanism to deter other organisms from eating the seeds. |
Plants are anchored in the ground and cannot run away when attacked, but they do not meekly allow themselves to be eaten! In many cases, plants that do not want to be eaten have developed complex chemical mechanisms that deter, distract, and even poison attackers. Many different plant poisons have been identified, but one of them is hydrogen cyanide - a poison that inhibits respiration. So far, more than 3000 species of plants have been identified that are capable of generating hydrogen cyanide to punish organisms that eat their leaves. The common apple is among these plants (Figure 1) [1]. These plants produce cyanide-containing molecules called cyanogenic compounds, for example, mandelonitrile (Figure 2).
Cyanogenic compounds (cyanogens) are inactive and generally not poisonous, so plants can produce and store them in special compartments inside their cells without causing themselves harm. In nearby but separate compartments, plants also store a special enzyme capable of transforming the inactive cyanogen into toxic hydrogen cyanide. Then, when insects or other animals bite down on the leaves of the plant, the compartments' walls are crushed, mixing the cyanogen with the enzyme and releasing toxic hydrogen cyanide right inside the attacker's mouth!
|
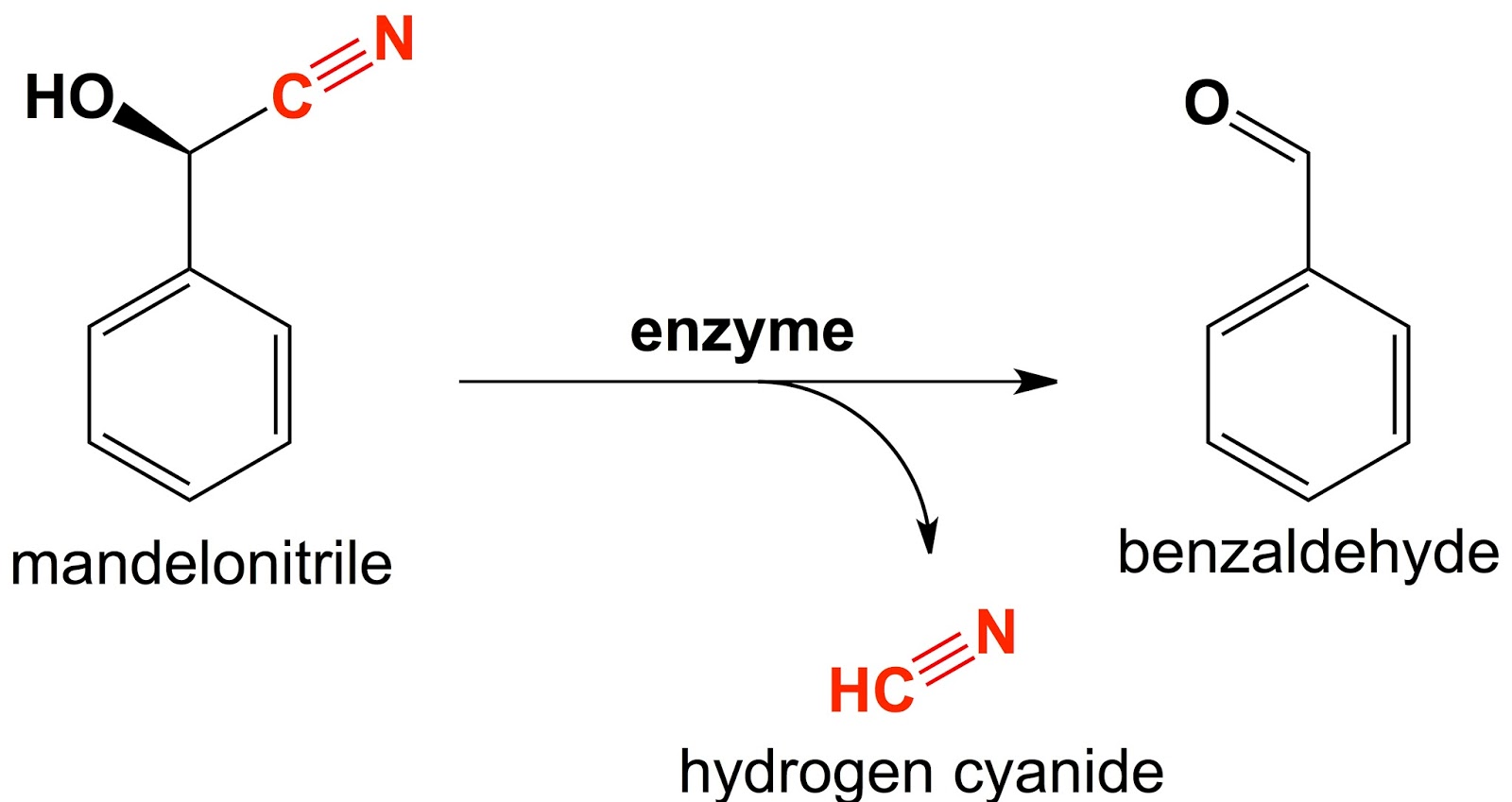
| Figure 2: Enzymatic activation of cyanogens. Mandelonitrile (a specific cyanogen) can be enzymatically converted into benzaldehyde (a side product) and active hydrogen cyanide. |
Plants produce and store cyanogens in many forms. Oftentimes they are stored linked with sugar molecules like glucose, in which case they are called cyanogenic glycosides. These are found in the seeds of many fruits in the rose family, like apples, plums, apricots, and peaches, but only in relatively small amounts. In small amounts, these seeds are not harmful to humans - so don't worry about eating a few apple seeds!
Cassava roots, a staple food in many African, Asian, and Latin American countries [2], contain substantial amounts of cyanogenic glycosides (Figure 3) [3]. Accordingly, cassava roots need to be processed before consumption. One method of doing this is to grind the cassava, exposing the activating enzyme to the cyanogenic glycosides, spreading the paste in a thin layer to allow it to dry. Once released, active hydrogen cyanide has a low boiling point, so much of it evaporates quickly and the cassava becomes safer to consume.
 |
| Figure 3: Cassava roots contain cyanogens that must be removed prior to human consumption. Grinding the roots and allowing active hydrogen cyanide to evaporate makes cassava safer to eat. This image is in the public domain. |
Cyanogens are currently being tested as alternatives to synthetic pesticides for specific applications. For example, approximately 25% of a wheat crop is lost to pests (rats, mice, insects, etc.) over the course of the agricultural process, with substantial losses occurring when the seeds are being stored prior to planting [4]. Synthetic pesticides are often applied to minimize these losses, but but we are learning that pesticides often have detrimental effects on nearby water sources and can be difficult to control when washed away by rain.
In response to the shortcomings of synthetic pesticides, materials chemists, inspired by cyanogens in nature, have invented a new coating that can applied to wheat seeds in storage. The seeds are coated with an enzyme, then a thin barrier, then a cyanogen [5]. When the barrier is ruptured by pests, the enzyme and cyanogen mix, releasing hydrogen cyanide. Such seeds are less prone to attack during the pre-planting phase of the agricultural process. While this application is still in developmental phases and is undergoing testing to determine its environmental and ecological effects, it is a step towards the protection of crops with naturally occurring, biodegradable materials.
Plant cyanogens are just one example of naturally occurring compounds that are useful to humans. The "2016 State of the World's Plants" report by the Royal Kew Gardens indicates that over 30,000 species of plants are useful to man. For example, over 5,000 feed us, 17,000 provide us with medicine, and 1,600 provide us with sources of fuel. Unstudied and unknown species hold chemical secrets that promise to aid humans in the future - these warrant study and exploration.
[1] Poulton, J.E., 1990. Cyanogenesis in plants. Plant Physiology, 94(2), pp.401-405.
[2] Food and Agriculture Organization. http://www.fao.org/ag/agp/agpc/gcds/
[3] Banea-Mayambu JP, Tylleskar T, Gitebo N, Matadi N, Gebre-Medhin M, Rosling H. (1997). Geographical and seasonal association between linamarin and cyanide exposure from cassava and the upper motor neurone disease konzo in former Zaire. Trop Med Int Health 2(12):1143-51]
[4] Encyclopedia of Pest Management, ed. D. Pimentel, CRC Press, 2002.
[5] Jonas G. Halter, Weida D. Chen, Nora Hild, Carlos A. Mora, Philipp R. Stoessel, Fabian M. Koehler, Robert N. Grass and Wendelin J. Stark (2014). Induced cyanogenesis from hydroxynitrile lyase and mandelonitrile on wheat with polylactic acid multilayer-coating produces self-defending seeds. J. Mater. Chem. A, 2: 853
